Clozapine N-oxide (CNO) dihydrochloride (Water soluble)
Product overview
| Name | Clozapine N-oxide (CNO) dihydrochloride (Water soluble) |
| Alternative names | CNO dihydrochloride, CNO.2HCl |
| Purity | >98% |
| Customer comments | We have been using Clozapine N-oxide (CNO) dihydrochloride (CNO.2HCl) from Hello Bio. We are fully satisfied with this product: it is readily water soluble and stable. We have administered CNO-2HCl to animals via IP injection and intracranial microinjection; and we have observed CNO-induced cellular and behavioral effects fully consistent with its expected activation of DREADD receptors." Verified customer, UTHSC Great product. Great company and product. Better pricing & similar quality compared to the most of the other companies. We used CNO dihydrochloride for both i.p. and drinking water delivery, and it worked fine. Nice service. Verified customer, University of Minnesota Works as expected, and significantly cheaper than alternatives. Will use this for future CNO purchases Verified customer, UCSF |
| Description | Dihydrochloride salt of CNO - the prototypical DREADD activator |
Images
Biological Data
| Biological description | Overview Clozapine N-oxide dihydrochloride (CNO) is the dihydrochloride salt of CNO which is the prototypical chemical DREADDs activator. It is a metabolite of clozapine. In rhesus macaques, CNO dihydrochloride shows improved bioavailability compared to CNO freebase with less conversion to clozapine
CNO activates the excitatory Gq- coupled DREADDs: hM3Dq, hM1Dq and hM5Dq (pEC50 values are 7.26 and 8.61 at hM3Dq and hM1Dq respectively). The hM3Dq DREADD is typically used for enhancing neuronal firing and activity (Gq- signaling in neuronal and non-neuronal cells). ‘Inhibitory’ (Gi- coupled) DREADDs: CNO also activates the inhibitory hM4Di and hM2Di Gi-coupled DREADDs (pEC50 = 6.89 at hM4Di). The hM4Di DREADD is the most commonly used inhibitory DREADD and is used for neuronal silencing. Gs and β-arrestin coupled DREADDs: CNO also activates the Gs- coupled DREADD (GsD) and the β-arrestin preferring DREADD: rM3Darr (Rq(R165L).
Care must therefore be taken in experimental design and proper controls should be incorporated, for example, the use of non-DREADD expressing animals may be appropriate (see Mahler and Aston-Jones (2018)).
Our stability studies have shown that CNO dihydrochloride is easier to solubilize and handle and have found that this product does not precipitate in solution unlike the freebase form of CNO (which, due to its inherent chemical properties requires careful handling and has been shown in the literature to precipitate in aqueous solution under certain conditions). For more info please see Stability of Water-Soluble DREADD ligands in Solution: A Technical Review and our handling guidelines below. |
| Application notes |
|
Solubility & Handling
| Storage instructions | -20°C (desiccate) |
| Solubility overview | Soluble in water (100 mM). Always store solutions at -20°C. |
| Handling | Storage of solid
Storage of solutions
Storage of solutions at room temperature
|
| Important | This product is for RESEARCH USE ONLY and is not intended for therapeutic or diagnostic use. Not for human or veterinary use |
Chemical Data
| Purity | >98% |
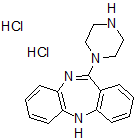
| Chemical name | 8-Chloro-11-(4-methyl-4-oxido-1-piperazinyl)-5H-dibenzo[b,e][1,4]diazepine dihydrochloride |
| Molecular Weight | 415.74 |
| Chemical structure |  |
| Molecular Formula | C18H19ClN4O.2HCl |
| CAS Number | 2250025-93-3 |
| SMILES | ClC(C=C1)=CC2=C1NC(C=CC=C3)=C3C(N4CC[N+](C)([O-])CC4)=N2.Cl.Cl |
| Source | Synthetic |
| InChi | InChI=1S/C18H19ClN4O.2ClH/c1-23(24)10-8-22(9-11-23)18-14-4-2-3-5-15(14)20-16-7-6-13(19)12-17(16)21-18;;/h2-7,12,20H,8-11H2,1H3;2*1H |
| InChiKey | MBRGKRXDVKTUPT-UHFFFAOYSA-N |
| Appearance | Orange solid |
References for Clozapine N-oxide (CNO) dihydrochloride (Water soluble)
-
A Comparative Study of the Pharmacokinetics of Clozapine N-Oxide and Clozapine N-Oxide Hydrochloride Salt in Rhesus Macaques.
Allen et al (2019) J Pharmacol Exp Ther 368(2) : 199-207 -
Pharmacokinetic and pharmacodynamic actions of clozapine-N-oxide, clozapine, and compound 21 in DREADD-based chemogenetics in mice.
Jendryka et al (2019) Sci Rep. 9(1) : 4522 -
DREADDs: The Power of the Lock, the Weakness of the Key. Favoring the Pursuit of Specific Conditions Rather than Specific Ligands.
Goutaudier et al (2019) eNeuro 6 : (5) -
CNO Evil? Considerations for the Use of DREADDs in Behavioral Neuroscience.
Mahler and Aston-Jones (2018) Neuropsychopharmacology doi: 10.1038 : /npp.2016.251 -
DREADD Agonist 21 Is an Effective Agonist for Muscarinic-Based DREADDs in Vitro and in Vivo
Thompson et al (2018) ACS Pharmacol. Transl. Sci. 10.1021 : /acsptsci.8b00012
-
Parabrachial opioidergic projections to preoptic hypothalamus mediate behavioral and physiological thermal defenses
Norris AJ et al (2021) Elife 10PubMedID: 33667158 -
Periaqueductal gray/dorsal raphe dopamine neurons contribute to sex differences in pain-related behaviors
Yu W et al (2021) Neuron 109(8) : 1365-1380.e5PubMedID: 33740416 -
Bi-directional regulation of cognitive control by distinct prefrontal cortical output neurons to thalamus and striatum
de Kloet SF et al (2021) Nat Commun 12(1) : 1994PubMedID: 33790281 -
Homeostatic synaptic scaling establishes the specificity of an associative memory
Wu CH et al (2021) Curr Biol 31(11) : 2274-2285.e5PubMedID: 33798429 -
Social isolation impairs the prefrontal-nucleus accumbens circuit subserving social recognition in mice
Park G et al (2021) Cell Rep 35(6) : 109104PubMedID: 33979617






 Understanding purity and quality - a guide for life scientists
Understanding purity and quality - a guide for life scientists