The store will not work correctly when cookies are disabled.
JavaScript seems to be disabled in your browser. For the best experience on our site, be sure to turn on Javascript in your browser.
Images (RS)-MCPG product vial image | Hello Bio
(RS)-MCPG product vial image | Hello Bio Biological Data Biological description Non-selective group I and II mGluR antagonist.
Also competitive antagonist of ACPD sensitive receptors.
Shows no effect on locomotor activity.
Water soluble (R,S)-MCPG sodium salt and (S)-MCPG sodium salt also available. (S)-MCPG also available.
Solubility & Handling Solubility overview Soluble in 0.1M NaOH (100mM)
Storage instructions Room temperature
Storage of solutions Prepare and use solutions on the same day if possible. Store solutions at -20°C for up to one month if storage is required. Equilibrate to RT and ensure the solution is precipitate free before use.
Shipping Conditions Stable for ambient temperature shipping. Follow storage instructions on receipt.
Important This product is for RESEARCH USE ONLY and is not intended for therapeutic or diagnostic use. Not for human or veterinary use.
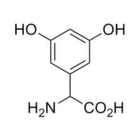
Chemical Data Chemical name (RS )-α-Methyl-4-carboxyphenylglycine
Chemical structure
Molecular Formula C10 H11 NO4
SMILES CC(C1=CC=C(C=C1)C(=O)O)(C(=O)O)N
InChi InChI=1S/C10H11NO4/c1-10(11,9(14)15)7-4-2-6(3-5-7)8(12)13/h2-5H,11H2,1H3,(H,12,13)(H,14,15)
InChiKey DNCAZYRLRMTVSF-UHFFFAOYSA-N
References for (RS)-MCPG References are publications that support the biological activity of the product
The metabotropic glutamate receptor antagonist (RS)-MCPG produces hyperlocomotion in amphetamine pre-exposed rats. Kim JH et al (1998) Neuropharmacology 37(2) : 189-97. MCPG antagonizes metabotropic glutamate receptors but not long-term potentiation in the hippocampus. Manzoni OJ et al (1994) Eur J Neurosci 6(6) : 1050-4. Competitive antagonism at metabotropic glutamate receptors by (S)-4-carboxyphenylglycine and (RS)-alpha-methyl-4-carboxyphenylglycine. Eaton SA et al (1993) Eur J Pharmacol 244(2) : 195-7.
Tell us about your publication! What Hello Bio product(s) have you cited?
Captcha Please type the letters and numbers below Submit
Non-selective group I and II mGluR antagonist
![(RS)-MCPG [146669-29-6] (RS)-MCPG [146669-29-6] Chemical Structure](https://cdn.hellobio.com/media/catalog/product//h/b/hb0037.png)





 Understanding purity and quality - a guide for life scientists
Understanding purity and quality - a guide for life scientists