Product overview
| Name | Rapamycin |
| Alternative names | Sirolimus |
| Purity | >98% |
| Description | mTOR inhibitor and potent immunosuppressant. Also used in inducible gene editing methods (e.g. CRISPR/Cas9) and promotes hPSC differentiation. |
Write Your Own Review
Biological Data
| Biological description | Overview Rapamycin is an mTOR inhibitor and potent immunosuppressant. It complexes with FKBP12 to bind mTORC1 to inhibit mTOR activity and block subsequent activation of p70 s6 kinase (IC50 = 50 pM). Active in vivo. Uses and applications Rapamcyin shows a variety of bioloigical actions. E.g it induces autophagy and apoptosis. Promotes hPSC differentiation to blood progenitor and mesendoderm cells. It can also be used in inducible CRISPR/Cas9 systems to enable inducible gene editing. Used as a chemical dimerizer in Chemically-inducible dimerization (CID). As mTOR is a central inflammation regulator, rapamycin has recently been investigated as part of cytokine storm / COVID-19 related research. |
Solubility & Handling
| Solubility overview | Soluble in ethanol (20 mM) and DMSO (50 mM) |
| Storage instructions | -20°C |
| Storage of solutions | Prepare and use solutions on the same day if possible. Store solutions at -20°C for up to one month if storage is required. Equilibrate to RT and ensure the solution is precipitate free before use. |
| Shipping Conditions | Stable for ambient temperature shipping. Follow storage instructions on receipt. |
| Important | This product is for RESEARCH USE ONLY and is not intended for therapeutic or diagnostic use. Not for human or veterinary use. |
Chemical Data
| Purity | >98% |
| Chemical name | (3S,6R,7E,9R,10R,12R,14S,15E,17E,19E,21S,23S,26R,27R,34aS)-9,10,12,13,14,21,22,23,24,25,26,27,32,33,34,34a-Hexadecahydro-9,27-dihydroxy-3-[(1R)-2-[(1S,3R,4R)-4-hydroxy-3-methoxycyclohexyl]-1-methylethyl]-10,21-dimethoxy-6,8,12,14,20,26-hexamethyl-23,27-epoxy-3H-pyrido[2,1-c][1,4]oxaazacyclohentriacontine-1,5,11,28,29(4H,6H,31H)-pentone |
| Molecular Weight | 914.18 |
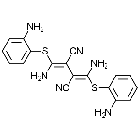
| Chemical structure | ![Rapamycin [ 53123-88-9 ] Rapamycin [ 53123-88-9 ] Chemical Structure](https://cdn.hellobio.com/media/catalog/product//h/b/hb2779.png) |
| Molecular Formula | C51H79NO13 |
| CAS Number | 53123-88-9 |
| PubChem identifier | 5284616 |
| SMILES | C[C@@H]1CC[C@H]2C[C@@H](/C(=C/C=C/C=C/[C@H](C[C@H](C(=O)[C@@H]([C@@H](/C(=C/[C@H](C(=O)C[C@H](OC(=O)[C@@H]3CCCCN3C(=O)C(=O)[C@@]1(O2)O)[C@H](C)C[C@@H]4CC[C@H]([C@@H](C4)OC)O)C)/C)O)OC)C)C)/C)OC |
| InChiKey | QFJCIRLUMZQUOT-HPLJOQBZSA-N |
| MDL number | MFCD00867594 |
| Appearance | White to off-white |
References for Rapamycin
References are publications that support the biological activity of the product
-
Targeting T-cell senescence and cytokine storm with rapamycin to prevent severe progression in COVID-19
Mahe et al (2020) Clin Immunol. 216 : 108464 -
A split-Cas9 architecture for inducible genome editing and transcription modulation.
Zetsche et al (2015) Nat Biotechnol 33(2) : 139-42 -
Chemical modulators of autophagy as biological probes and potential therapeutics.
Flemin et al (2011) Nat Chem Biol 7(1) : 9-17 -
Rapamycin, a specific inhibitor of the mammalian target of rapamycin, suppresses lymphangiogenesis and lymphatic metastasis.
Kobayashi et al (2007) Cancer Sci 98(5) : 726-33 -
Rapamycins: mechanism of action and cellular resistance.
Huang et al (2003) Cancer Biol Ther 2(3) : 222-32
Publications
These publications cite the use of Rapamycin purchased from Hello Bio:
-
The Effects of Rapamycin on the Intestinal Graft in a Rat Model of Cold Ischemia Perfusion and Preservation.
Caleb I et al (2022) Metabolites 12PubMedID: 36144199 -
Characterizing autophagy in the cold preservation of small bowel grafts
Caleb I, (2022) Thesis -
Characterizing Autophagy in the Cold Ischemic Injury of Small Bowel Grafts: Evidence from Rat Jejunum
Caleb I et al (2021) Metabolites 11(6)PubMedID: 34204418 -
NMDA receptor partial agonist GLYX-13 alleviates chronic stress-induced depression-like behavior through enhancement of AMPA receptor function in the periaqueductal gray
Yang PS et al (2020) Neuropharmacology 178 : 108269PubMedID: 32791085





 Understanding purity and quality - a guide for life scientists
Understanding purity and quality - a guide for life scientists