JHU37160 (DREADD ligand)
Certificate of Analysis
Product overview
| Name | JHU37160 (DREADD ligand) |
| Alternative names | J60 |
| Purity | >98% |
| Description | Novel DREADD agonist with high affinity and potency for hM3Dq and hM4Di. Active in vivo. Freebase. |
Images
Biological Data
| Biological description | Overview JHU37160 is reported to be a novel DREADD agonist with high in vivo DREADD potency for CNS applications. It has high affinity in vitro for hM3Dq and hM4Di (Ki values are 1.9 nM (hM3Dq) and 3.6 nM (hM4Di). It selectively displaces [3H]clozapine from DREADDs and not from other clozapine-binding sites at concentrations up to 10 nM when tested for in situ [3H]clozapine displacement in brain tissue from WT and D1-DREADD mice. JHU37160 activates hM3Dq and hM4Di with high potency and efficacy in fluorescent and BRET-based assays in HEK-293 cells (EC50 values are 18.5 and 0.2 nM at hM3Dq and hM4Di respectively. Occupancy JHU37160 exhibits high in vivo DREADD occupancy and was not reported to be a P-gp substrate. In vivo application JHU37160 is reported to be a potent in vivo DREADD agonist, which selectively inhibits locomotor activity in D1-hM3Dq and D1-hM4Di mice without any significant locomotor effects observed in wild type (WT) mice (at doses ranging 0.01 - 1 mg/kg). It also produces robust and selective increases in hM3Dq-stimulated locomotion in rats expressing hM3Dq in tyrosine hydroxylase expressing neurons (at doses ranging 0.01 – 0.3 mg/kg). In in vivo electrophysiology experiments in mice, JHU37160 produces rapid and potent hM4Di-driven inhibition of light-evoked neuronal activation (at a dose of 0.1 mg/kg). While its selectivity is not ideal (i.e. comparable to clozapine), its high in vivo potency allows for dose adjustments with minimal off-target effects. The compound exhibits promising characteristics for DREADD use in monkeys. Hydrochloride form recently shown to have dose-dependent anxiogenic effects in male Wistar and Long-Evans rats, regardless of DREADD expression, with no impact on locomotor behvaiour suggesting that high doses (e.g. 1mg/kg) should be avoided in chemogenetic experiments designed to evaluate circuit manipulation on anxiety-like behavior in rats. Water soluble version also available. Sold under license from the NIH, US patent pending 62/627,527 |
Solubility & Handling
| Solubility overview | Soluble in DMSO (100 mM) |
| Storage instructions | Room temperature |
| Storage of solutions | Prepare and use solutions on the same day if possible. Store solutions at -20°C for up to one month if storage is required. Equilibrate to RT and ensure the solution is precipitate free before use. |
| Shipping Conditions | Stable for ambient temperature shipping. Follow storage instructions on receipt. |
| Important | This product is for RESEARCH USE ONLY and is not intended for therapeutic or diagnostic use. Not for human or veterinary use |
Chemical Data
| Purity | >98% |
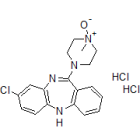
| Chemical name | 8-chloro-11-(4-ethylpiperazin-1-yl)-4-fluoro-5H-dibenzo[b,e][1,4]diazepine |
| Molecular Weight | 358.84 |
| Chemical structure |  |
| Molecular Formula | C19H20ClFN4 |
| CAS Number | 2369979-68-8 |
| SMILES | CCN1CCN(CC1)C3=Nc4cc(Cl)ccc4Nc2c3cccc2F |
| Source | Synthetic |
| InChi | InChI=1S/C19H20ClFN4/c1-2-24-8-10-25(11-9-24)19-14-4-3-5-15(21)18(14)22-16-7-6-13(20)12-17(16)23-19/h3-7,12,22H,2,8-11H2,1H3 |
| InChiKey | SWSCWOSASZXIRK-UHFFFAOYSA-N |
| Appearance | Yellow solid |
| Licensing details | Sold under license from the NIH, US patent pending 62/627,527 |
References for JHU37160 (DREADD ligand)
-
High dose administration of DREADD agonist JHU37160 produces increases in anxiety-like behavior in male rats.
Van Savage J et al (2023) Behavioural brain research 452 : 114553 -
High-potency ligands for DREADD imaging and activation in rodents and monkeys.
Bonaventura et al (2019) Nat Commun. 10(1) : 4627 -
0067 Humanized Chemogenetic Approach to Treat Sleep Apnea
Curado et al (2019) Sleep (42) : A28 -
OP-01-02 Graft-host synaptic connectivity can be chemogenetically inhibited with clinically relevant activators to eliminate graft-induced dyskinesias (GID) without loosing anti-parkinsonian benefits of dopaminergic grafts
Subramanian et al (2019) World Congress On Parkinson's Disease And Related Disorders 2019 : Poster Abstract -
DREADDs: The Power of the Lock, the Weakness of the Key. Favoring the Pursuit of Specific Conditions Rather than Specific Ligands.
Goutaudier et al (2019) eNeuro 6 : (5)






 Understanding purity and quality - a guide for life scientists
Understanding purity and quality - a guide for life scientists