Clozapine N-Oxide (freebase) - a technical review on stability, solubility and use in the lab
 Introduction
Introduction
Background
Working with CNO (freebase) as a solid
- What should CNO (freebase) look like?
- What temperature should I store the solid compound at?
- Is the solid powder light sensitive?
- Is the solid powder air sensitive?
- How do I weigh the compound to make my stock solution?
Solubilizing / dissolving CNO (freebase)
- How do I solubilize CNO (freebase) to make my stock solution? Which solvent / buffer can I use?
- What should I do if I find precipitate in my stock solution?
- What concentration should my stock solution be?
- What should my stock solution look like?
- Is there a recommended way to solubilize CNO (freebase)?
Storing CNO (freebase)
- Can I store my stock solution of Clozapine N- oxide (CNO (freebase))?
- How long does Clozapine N-oxide (CNO freebase) stay stable in solution?
- Is my stock solution light sensitive?
- Is my stock solution air sensitive?
Using CNO (freebase) in the laboratory
- Which DREADDs does CNO activate?
- How do I express DREADDs?
- How can I administer CNO?
- Which buffers / solvents are used to administer CNO?
- What doses of CNO are used?
- Can CNO be given in drinking water or chow?
- Does CNO taste bitter?
- Administration via minipump
- Does CNO have off-site effects?
- Are there any other DREADD ligands?
- Can DREADDs be used together?
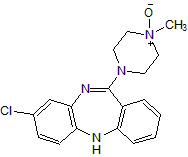
Figure 1 – Chemical structure
of Clozapine N-oxide (CNO). Clozapine N-oxide is an
oxidized piperazine substituted benzodiazepine and a
metabolite of the antipsychotic compound Clozapine
Introduction
DREADDs are an increasingly popular chemogenetic tool which allows the control of activity of a wide range of cell types (Find out more in our mini-review).
DREADDs are engineered to be activated by synthetic small molecules (designer drugs) such as Clozapine N-Oxide (CNO) which is an oxidized piperazine substituted benzodiazepine and a metabolite of the antipsychotic compound Clozapine. CNO is the most widely used compound to activate DREADDs such as the excitatory hM3Dq DREADD and the inhibitory hM4Di DREADD.
This technical review aims to answer common questions on how to handle, store and use Clozapine N-oxide (CNO) (freebase) for optimal results, and includes data from stability and solubility studies, using CNO (freebase) manufactured by Hello Bio.
For information regarding the stability and solubility of other DREADD ligands (including water soluble clozapine N-oxide dihydrochloride and Compound 21 (DREADD agonist 21) dihydrochloride) please see: Stability of Water-Soluble DREADD Ligands in Solution: A Technical Review.
Background
Hello Bio manufactures high purity (>99%), CNO freebase and makes it available to researchers at prices around 50% less than other life science suppliers that offer this product.
We have found that the appearance and solubility of CNO from other suppliers may differ due to differing manufacturing procedures. CNO from other suppliers may also contain alcohol impurities such as ethanol/methanol.
A review of the available literature indicates that CNO should be handled, used and stored carefully. Following our manufacture of CNO (freebase), we have undertaken extensive chemical, stability and solubility studies of this compound to provide a detailed methodology and handling protocol, and to answer questions that researchers may have.
Please note that throughout this review we have used the nomenclature CNO (freebase). The handling and storage information outlined in this review is specific to CNO (freebase) as manufactured by Hello Bio.
Working with CNO (freebase) as a solid

Figure 2 – Picture of solid CNO (freebase)
What should CNO (freebase) look like?
CNO (freebase) is an amorphous, yellow powder (see Fig 2).
What temperature should I store the solid compound at?
We undertake regular quality control analysis of the solid compound and have found no deterioration in product purity when stored at room temperature.
We therefore recommend that you store solid CNO (freebase) powder from Hello Bio at room temperature (desiccate).
Is the solid powder light sensitive?
No - we have found no evidence that Hello Bio CNO (freebase) is sensitive to light.
Is the solid powder air sensitive?
No - we have found no evidence that Hello Bio CNO (freebase) is sensitive to air.
How do I weigh the compound to make my stock solution?
You should weigh out the quantity of product that you require for your experiment as the amount of product in our vials isn’t weighed out accurately enough for direct addition of solution.
Solubilizing / dissolving CNO (freebase)
How do I solubilize CNO (freebase) to make my stock solution? Which solvent / buffer can I use?
Due to the inherent chemical properties of CNO, this product requires careful handling – our guidelines are detailed below.
Batch to batch variation in solubility can occur - some batches are water soluble but ongoing solubility in aqueous solutions can be unpredictable. CNO has been shown in the literature to precipitate in solution and following further solubility studies that we have undertaken, we have found that precipitation occurred in some concentrated (100mM) samples of CNO dissolved in water (precipitation was observed in 20% of 100 mM solutions of CNO (dissolved in water) which were stored for four days. This precipitation was readily reversed by warming in a water bath at 40° (see Table 1)).
We therefore recommend using DMSO when working with CNO freebase. If you need to work with aqueous solutions, you should use CNO dihydrochloride.
Solubilizing in DMSO
CNO (freebase) is soluble in DMSO to 100 mM.
Storage of solutions in DMSO
- Aliquot out the solution into tightly sealed vials for storage.
- Storage of solutions should be at room temperature.
- Please take care to ensure that your product is completely dissolved in your solution before use.
Solubilizing CNO (freebase) in aqueous solutions (e.g. water / saline)
We do not recommend using CNO freebase with aqueous solutions however, we provide these guidelines if you do:
- Make up solutions and use immediately. Do not store solutions.
- Ensure you work in a dust free environment when preparing and handling solutions.
- Ensure that the product and solvents are at ambient temperature before preparing solutions. This means that you should allow the product to equilibrate to RT for at least one hour before opening and using.
- Please take care to ensure that your product is completely dissolved in your solution before use.
In line with these findings, different groups also recommend that when possible CNO solutions should be used on the same day; Wirtshaftern and Stratford (2015) state that “The CNO was prepared immediately before use as it tended to precipitate if stored” [1].
Stability in solution
Our stability studies have shown that CNO in solution remains chemically stable (>99% purity by HPLC) for at least 4 weeks at room temperature.
Storage of aqueous solutions
We recommend that you do not store aqueous solutions (e.g. water/saline) of CNO. However, if this is necessary:
- Aliquot out the solution into tightly sealed vials for storage. Storage of solutions should be at room temperature.
- Do not store solutions at temperatures below room temperature (i.e. +4°C/ -20°C) - this is more likely to lead to precipitation of the compound.
- Preferably, solutions should be made in transparent, colorless vials so that any precipitation can be observed and remedied prior to use.
- Always check that your product is completely dissolved before use; if precipitation is found follow the steps below.
Precipitation
If you find precipitate in your solution, gently heat your solution in a water bath to approx 40°C and the compound should readily re-dissolve. Always take care to ensure that the compound is completely dissolved before use.
| Buffer/solvent | Concentration (mM unless otherwise stated) |
Temperature (°C unless otherwise stated) |
Test duration (days) | Result |
| Saline | 100 | RT | - | Soluble |
| PBS | 100 | RT | - | Soluble |
| Water | 100 | RT | 7 | Soluble* |
| Water | 1mg/ml | RT | 7 | Soluble |
| Water | 20 | RT | 7 | Soluble |
| Water | 50 | RT | 7 | Soluble |
| Water | 100 | RT | 7 | Soluble |
| Water | 20 | 37 | 7 | Soluble |
| Water | 50 | 37 | 7 | Soluble |
| Water | 100 | 37 | 7 | Soluble |
| Water | 1mg/mL | 4 | 7 | Soluble |
| Water | 20 | 4 | 7 | Soluble |
| Water | 50 | 4 | 7 | Soluble |
| Water | 100 | 4 | 7 | Soluble* |
| DMSO | 50 | freezing overnight then rethawed, then RT |
7 | Soluble |
| DMSO | 100 | freezing overnight then rethawed, then RT |
7 | Soluble |
| DMSO | 50 | RT | 7 | Soluble |
| DMSO | 100 | RT | 7 | Soluble |
* After 4 days, precipitation was observed in 20% of CNO samples dissolved in water (100 mM). Precipitation was readily reversed by warming in a water bath at 40°C.
What should I do if I find precipitate in my stock solution?
Gently heat your solution in a water bath to approx 40°C and the compound should readily re-dissolve.
Always take care to ensure that the compound i scompletely dissolved before use.
What concentration should my stock solution be?
This depends on your experiment and the working concentration you require.
Please see Table 3 for same examples of different concentrations reported in various papers.
What should my stock solution look like?
When dissolved in water, saline or PBS your solution of CNO (freebase) should be a clear, yellow to orange color (depending on the concentration) – see Fig 3.
Figure 3 - Hello Bio CNO (freebase) dissolved in water at 20, 50 and 100mM
Similarly, when dissolved in DMSO, your solution should also be a clear, yellow to orange color (depending on the concentration) – see Fig 4.
Figure 4 - Hello Bio CNO (freebase) dissolved in DMSO at 50 and 100mM
Is there a recommended way to solubilize CNO (freebase)?
Various groups have different approaches to solubilizing CNO. For example, DMSO is commonly used to dissolve CNO before addition to saline for injection.
Smith et al (2016) describe some examples of different group’s approaches on page 13 [2].
The approach you choose depends on your experimental requirements.
Storing CNO (freebase)
Can I store my stock solution of Clozapine N- oxide (CNO (freebase))?
Storage of solutions in DMSO
- Aliquot out the solution into tightly sealed vials for storage.
- Storage of solutions should be at room temperature.
- Please take care to ensure that your product is completely dissolved in your solution before use.
We recommend that you do not store aqueous solutions (e.g. water/saline) of CNO. However, if this is necessary:
- Aliquot out the solution into tightly sealed vials for storage. Storage of solutions should be at room temperature.
- Do not store solutions at temperatures below room temperature (i.e. +4°C/ -20°C) - this is more likely to lead to precipitation of the compound.
- Preferably, solutions should be made in transparent, colorless vials so that any precipitation can be observed and remedied prior to use.
- Always check that your product is completely dissolved before use; if precipitation is found follow the steps below.
Precipitation
If you find precipitate in your solution, gently heat your solution in a water bath to approx 40°C and the compound should readily re-dissolve. Always take care to ensure that the compound is completely dissolved before use.
How long does Clozapine N-oxide (CNO freebase) stay stable in solution?
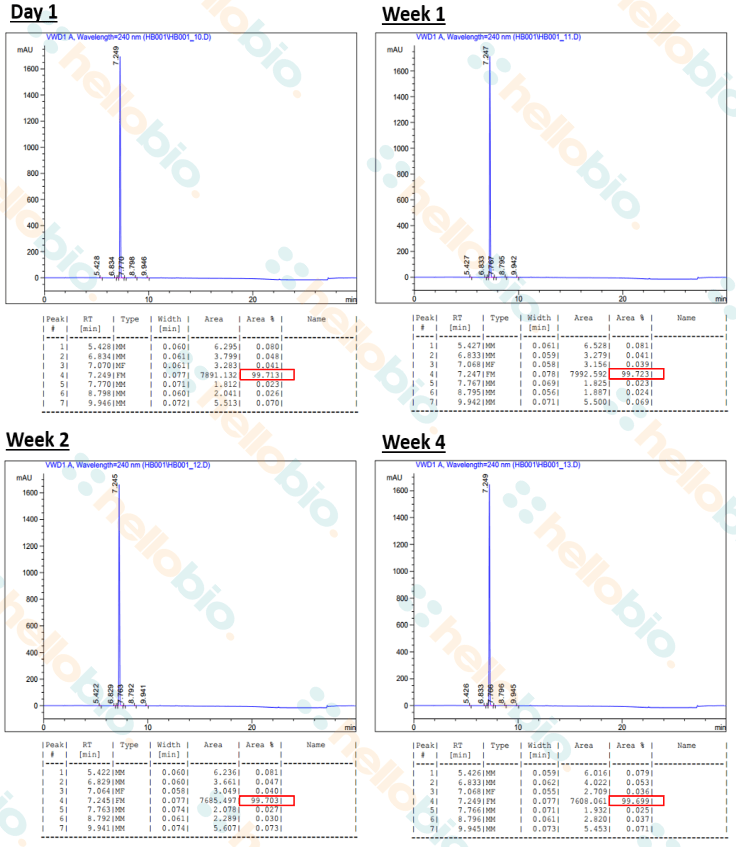
Stability studies were performed on 100 mM solutions of Clozapine N-oxide dissolved in water, which were kept at room temperature and ambient lighting for 4 weeks. No special precautions to exclude air were taken.
HPLC analysis of samples at day 1, week 1, week 2 and week 4 showed no deterioration in product purity (>99.60%) (see Fig 5 / Table 2).
Our studies therefore show that CNO (freebase) is chemically stable in solution at room temperature for at least 4 weeks.
Figure 5 - HPLC analysis of 100 mM CNO solutions dissolved in water after 4 weeks of storage at RT and at ambient lighting. A purity of >99.6% was confirmed after 1 day, 1 week, 2 weeks and 4 weeks of storage, confirming that no deterioration in product purity occurred over 4 weeks.
Purity was determined by HPLC analysis using reversed phase chromatography on a C18 column, using a TFA-acetonitrile/TFA-water gradient and detecting at 240 nm.
| Time (weeks) | Purity |
| 0 | >99.6% |
| 1 | >99.6% |
| 2 | >99.6% |
| 4 | >99.6% |
|
Table 2 - Purity of CNO solutions dissolved in water (100 mM) after |
|
Is my stock solution light sensitive?
No - we have found no evidence that Hello Bio CNO (freebase) is light sensitive.
Is my stock solution air sensitive?
No - we have found no evidence that Hello Bio CNO (freebase) is air sensitive.
Using CNO (freebase) in the laboratory
Based on a review of the literature, the following is a summary of useful information relating to the usage of Clozapine N-oxide (CNO) (freebase). Do you have any questions that aren’t listed below? If so, contact us on technicalhelp@hellobio.com and we’ll try our very best to help!
Which DREADDs does CNO activate?
CNO activates various DREADDs including the excitatory Gi-DREADDs (hM3Dq, hM1Dq and hM5Dq), the inhibitory Gq- DREADDs (hM4Di, hM2Di), the Gs -DREADD (GsD) and the β-arrestin preferring DREADD rM3Darr(Rq(R165L)).
How do I express DREADDs?
DREAADs can be selectively expressed in a targeted cell population by using viral vectors or transgenic mice generated by random or site-specific targeting [3].
How can I administer CNO?
There is some debate in the literature regarding the brain penetrant properties of CNO – some groups report high BBB penetration [4], whilst a recent study suggests that CNO systemically administered does not readily cross the blood-brain-barrier in vivo, and converts to clozapine which activates DREADDs [5]. Care must be taken in experimental design and proper controls should be incorporated
CNO has been commonly administered via injection (e.g. intraperitoneal injection [6,7,8,9], subcutaneous [10], or directly infused intracranially [12,13]). It has been administered via drinking water [14], chow [15] and recently osmotic pump [16].
Please see Table 3 for some examples of different administration routes used in various papers.
Which buffers / solvents are used to administer CNO?
Please consult the literature to choose the most appropriate administration buffer for your experiment.
Sterile saline is most commonly used for i.p. injection. Water, PBS and ACSF have also been used.
CNO has also been added to recording buffer in patch clamp electrophysiology experiments.
Please see Table 3 for same examples of buffers / solvents used in various papers.
What doses of CNO are used?
The dose of CNO administered in the literature varies and a wide range of doses have been used e.g. 0.2 – 10 mg/kg [7]. We recommend consulting the literature to ascertain the appropriate dose for your experiment.
Please see Table 3 for same example doses used in various papers.
Can CNO be given in drinking water or chow?
CNO has been delivered by food or water [3]. This can be useful for long-term studies and if you want to avoid repeated handling and/or injection of animals [14].
Please see Table 3 for same example papers which given CNO via drinking water [17,18,15].
Does CNO taste bitter?
According to Bryan Roth’s blog a “A small amount of saccharine in the drinking water will mask the slightly bitter taste of CNO” [33]. Milosavljevic et al (2016) mention that their animals were “presented with 0.25 mg/ml CNO in sweetened drinking water (0.2% saccharine and 4% sucrose)” [17].
Administration via minipump
There are limited citations using CNO via osmotic pump. However, Donato et al (2017) used an osmotic minipump implanted subcutaneously on mice pup's backs to deliver CNO (100 µl of CNO solution (1mg/ml) with a flow of 0.5 µl per hour for up to 7 days [16].
A thesis by Mason, X et al (2015) also used an osmotic pump to deliver CNO (diluted in 0.9% sterile saline to a concentration of 5mg/mL). Approx 240 µL of CNO solution was loaded into a subcutaneously implanted Alzet® 2004 pump [19].
A common question is how long does CNO stay stable in solution? As mentioned above, our stability studies showed that CNO was chemically stable in solution at room temperature for at least 4 weeks. HPLC analysis during this time showed no deterioration in product purity.
Does CNO have off-site effects?
Clozapine N-oxide lacks appreciable affinity (Ki = 1 µM) at relevant native CNS targets [2]. However, CNO is known to back-metabolize to clozapine in humans, guinea pigs [2], primates and mice [5].
Recent studies have demonstrated that CNO may exert behavioural effects on rats that do not express DREADDs [7] and Salomon et al (2016) have also demonstrated biological activity of DREADDs in the absence of CNO [23].
Gomez et al (2017) have reported that CNO has a low affinity for DREADDs in comparison with clozapine, and that systemically administered CNO does not readily cross the blood-brain-barrier in vivo, and converts to clozapine which activates DREADDs [5].
Maclaren et al (2016) recommend that all experiments which utilize CNO should verify that the administration of CNO does not have effects in the species and strain being used. Importantly, they emphasize the need for a CNO-only DREADD-free control group when designing DREADD-based experiments [7].
Care must therefore be taken in experimental design and proper controls should be incorporated.
Are there any other DREADD ligands?
Yes – the clozapine analog Compound 21 (which is also available in a water soluble form) acts as a highly potent hM3Dq agonist. There are indications that Compound 21 may not be metabolized to clozapine [22] and may have equivalent potency in vivo when compared with CNO.
Perlapine and the water-soluble perlapine dihydrochloride are novel, potent and selective hM3Dq DREADD receptor agonists and are better suited to translational studies [22]. Additionally, the inert ligand Salvinorin B (SALB) acts at the inhibitory KORD DREADD [3].
For information regarding the stability and solubility of other DREADD ligands (including water soluble clozapine N-oxide dihydrochloride and Compound 21 (DREADD agonist 21) dihydrochloride) please see: Stability of Water-Soluble DREADD Ligands in Solution: A Technical Review.
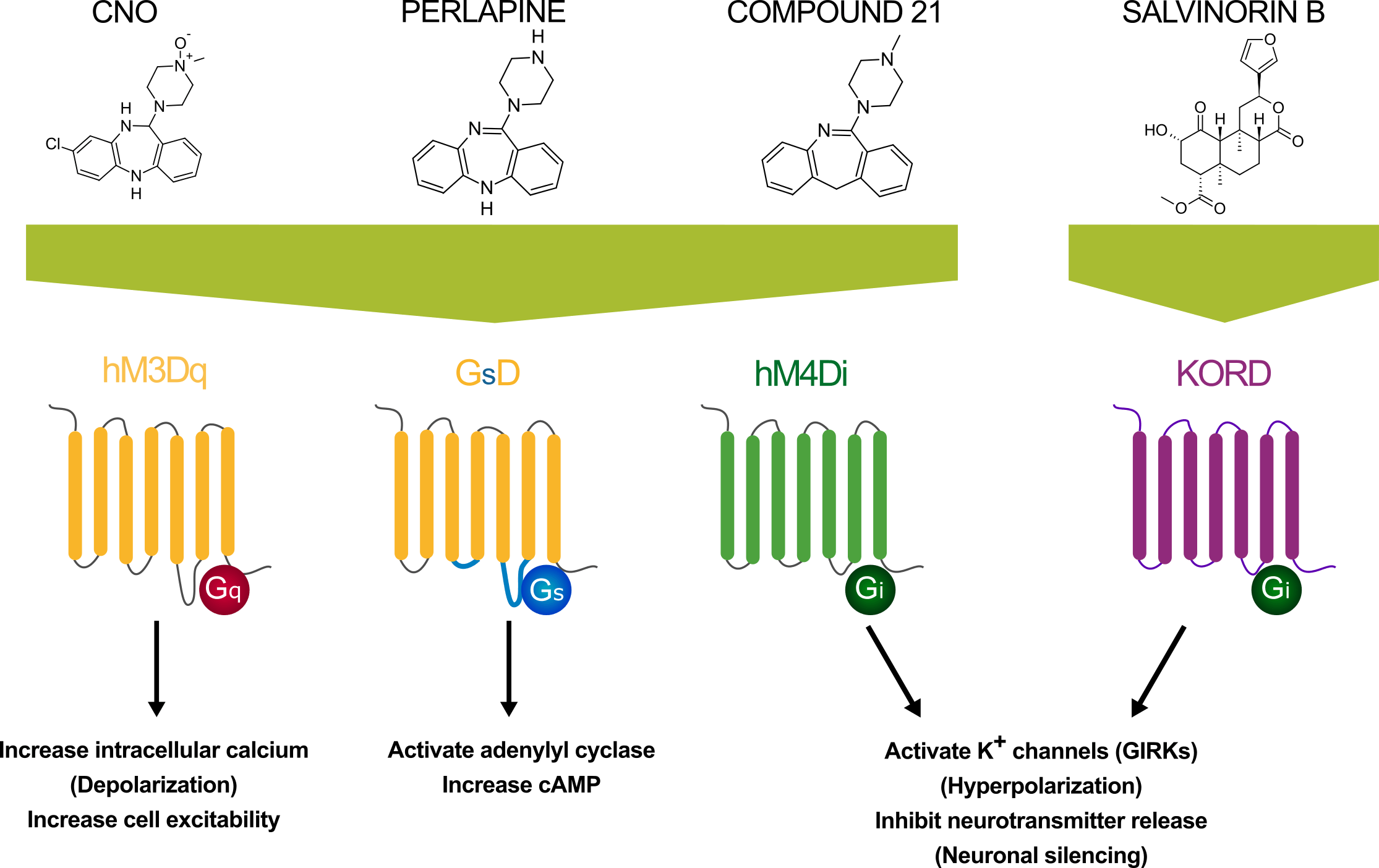
Figure 6 – Different designer compounds (CNO, perlapine, Compound 21 and SALB) are used to activate different DREADDs.
Can DREADDs be used together?
Yes - due to the existence of multiple DREADDs which can be activated by different ligands, DREADDs can be used in a ‘multiplexed’ approach in which DREADDs which respond to CNO (e.g. hM3Dq / hM4Di) and the SALB responsive KORD DREADD can be expressed in the same animal to enable bidirectional control of neuronal activity [27, 24, 3, 25].
| Reference | In vitro or in vivo usage? | Administration method / usage | Dosage (mg/kg) | Administration vehicle | Final concentration | Solvent used for stock solution | Final DMSO concentration | Stock concentration (mg/ml) |
| 21 | In vitro | Electrophysiology | Recording buffer | 10uM | DMSO | 100mM | ||
| 19 | In vitro | Electrophysiology | Saline | 500nM - 1uM | ||||
| 24 | In vitro | Electrophysiology | 10uM | |||||
| 6 | In vitro | Electrophysiology | 30uM | Extracellular solution | ||||
| 25 | In vitro | Electrophysiology | 10 | N/A | 1uM | Saline | ||
| 16 | In vitro | 1uM | ||||||
| 15 | In vivo | Drinking water | 5-6.25 | Water (0.2% saccharine and 4% surcose added) | ||||
| 16 | In vivo | Drinking water | 0.25mg/ml | Water | ||||
| 13 | In vivo | Drinking water | 0.25mg/ml | Water | ||||
| 24 | In vivo | Drinking water | 5 | Water | 40mg/l | Water | ||
| 11 | In vivo | Intracranial injection | Saline | 3 μM | Saline | |||
| 1 | In vivo | Intracranial injection | 2.5 | Water | 2.5 & 10mg/ml | DMSO | 20% | |
| 10 | In vivo | Intracranial injection | ACSF | 100uM & 1mM | ||||
| 25 | In vivo | Intraperitoneal (i.p.) | 10 | Saline | 1mg/ml | Saline | ||
| 5 | In vivo | Intraperitoneal (i.p.) | 1 | Saline | 1, 2 and 5 mg/ml | DMSO | 0.50% | |
| 15 | In vivo | Intraperitoneal (i.p.) | 5 | |||||
| 16 | In vivo | Intraperitoneal (i.p.) | ||||||
| 13 | In vivo | Intraperitoneal (i.p.) | 2 | PBS | 0.2mg/ml | PBS | ||
| 9 | In vivo | Intraperitoneal (i.p.) | 3 | Saline | DMSO | 0.50% | ||
| 21 | In vivo | Intraperitoneal (i.p.) | 3 | Saline | DMSO | 100 mM | ||
| 18 | In vivo | Intraperitoneal (i.p.) | 1 | |||||
| 26 | In vivo | Intraperitoneal (i.p.) | 1 | Saline | Saline | |||
| 23 | In vivo | Intraperitoneal (i.p.) | 3 | Saline | Saline | |||
| 19 | In vivo | Intraperitoneal (i.p.) | 0.1, 0.3, 0.5, 1 & 5 | Saline | ||||
| 24 | In vivo | Intraperitoneal (i.p.) | 2 & 5 | |||||
| 6 | In vivo | Intraperitoneal (i.p) | 1 | |||||
| 10 | In vivo | Intraperitoneal (i.p.) | 0.1, 1, 10 & 20 | Saline | DMSO | 5% | 1ml/kg | |
| 27 | In vivo | Intraperitoneal (i.p.) | 1 | |||||
| 28 | In vivo | Intraperitoneal (i.p.) | 0.1, 1 and 10 | Saline | 5uM | 2.50% | 10mg/ml | |
| 17 | In vivo | Osmotic minipump | ||||||
| 14 | In vivo | Osmotic minipump | Saline | 5mg/ml | ||||
| 8 | In vivo | Subcutaneous (s.c.) | 3 & 10 | PBS | 10mg/ml | DMSO | 15% | 66mg/ml |
| 29 | In vivo | Subcutaneous (s.c.) | 1 | Saline |
References
- Wirtshafter D et al (2016) Chemogenetic inhibition of cells in the paramedian midbrain tegmentum increases locomotor activity in rats. Brain Res 1632 98-106. Pubmed ID:26707405
- Smith KS et al (2016) DREADDs: Use and application in behavioral neuroscience. Behav Neurosci 130(2) 137-55. Pubmed ID:26913540
- Whissell PD et al (2016) The Use of DREADDs to Deconstruct Behavior. Front Genet 7 70 Pubmed ID:27242888
- Roth B et al (2016) DREADDs for Neuroscientists. Neuron 89(4):683-94 Pubmed ID: 26889809
- Gomez et al (2017) Chemogenetics revealed: DREADD occupancy and activation via converted clozapine. Science 357(6350):503-507 Pubmed ID: 28774929
- Wess J et al (2013) Novel designer receptors to probe GPCR signaling and physiology. Trends Pharmacol Sci 34(7) 385-92. Pubmed ID:23769625
- MacLaren DA et al (2016) Clozapine N-Oxide Administration Produces Behavioral Effects in Long-Evans Rats: Implications for Designing DREADD Experiments. eNeuro 3(5) Pubmed ID:27822508
- Inutsuka A et al (2014) Concurrent and robust regulation of feeding behaviors and metabolism by orexin neurons. Neuropharmacology 85 451-60. Pubmed ID:24951857
- Xie AX et al (2015) Molecular approaches for manipulating astrocytic signaling in vivo. Front Cell Neurosci 9 144 Pubmed ID:25941472
- Raper J et al (2017) Metabolism and Distribution of Clozapine-N-oxide: Implications for Nonhuman Primate Chemogenetics. ACS Chem Neurosci Pubmed ID:28324647
- Scofield MD et al (2015) Gq-DREADD Selectively Initiates Glial Glutamate Release and Inhibits Cue-induced Cocaine Seeking. Biol Psychiatry 78(7) 441-51. Pubmed ID:25861696
- Mahler SV(2014) Designer receptors show role for ventral pallidum input to ventral tegmental area in cocaine seeking. 17(4):577-85. Pubmed ID:24584054
- Stachniak TJ et al (2014) Chemogenetic synaptic silencing of neural circuits localizes a hypothalamus midbrain pathway for feeding behavior. Neuron 82(4) 797-808. Pubmed ID:24768300
- Spangler SM et al (2017) Optogenetic approaches for dissecting neuromodulation and GPCR signaling in neural circuits. Curr Opin Pharmacol 32 56-70. Pubmed ID:27875804
- Carvalho Poyraz F et al (2016) Decreasing Striatopallidal Pathway Function Enhances Motivation by Energizing the Initiation of Goal-Directed Action. J Neurosci 36(22) 5988-6001. Pubmed ID:27251620
- Donato F et al (2017) Stellate cells drive maturation of the entorhinal-hippocampal circuit. Science 355(6330) Pubmed ID:28154241
- Milosavljevic N et al (2016) Chemogenetic Activation of Melanopsin Retinal Ganglion Cells Induces Signatures of Arousal and/or Anxiety in Mice. Curr Biol 26(17) 2358-63. Pubmed ID:27426512
- Jain S et al (2013) Chronic activation of a designer G(q)-coupled receptor improves beta cell function. J Clin Invest 123(4) 1750-62. Pubmed ID:23478411
- Mason, Xenos (2015) Neural Circuit Mechanisms Underlying the Exacerbation of Alzheimer’s Disease by Chronic Stress Doctoral dissertation, Harvard medical school http://nrs.harvard.edu/urn-3:HUL.InstRepos:15821598
- Guettier JM et al (2009) A chemical-genetic approach to study G protein regulation of beta cell function in vivo. Proc Natl Acad Sci U S A 106(45) 19197-202. Pubmed ID:19858481
- Alexander GM et al (2009) Remote control of neuronal activity in transgenic mice expressing evolved G protein-coupled receptors. Neuron 63(1) 27-39. Pubmed ID:19607790
- Chen X et al (2015) The first structure-activity relationship studies for designer receptors exclusively activated by designer drugs. ACS Chem Neurosci 6(3) 476-84. Pubmed ID:25587888
- Saloman JL et al (2016) Gi-DREADD Expression in Peripheral Nerves Produces Ligand-Dependent Analgesia, as well as Ligand-Independent Functional Changes in Sensory Neurons. J Neurosci 36(42) 10769-10781. Pubmed ID:27798132
- Vardy E et al (2015) A New DREADD Facilitates the Multiplexed Chemogenetic Interrogation of Behavior. Neuron 86(4) 936-46. Pubmed ID:25937170
- Rapanelli M et al (2017) Histamine modulation of the basal ganglia circuitry in the development of pathological grooming. Proc Natl Acad Sci U S A 114(25) 6599-6604. Pubmed ID:28584117
- Urban DJ et al (2016) Elucidation of The Behavioral Program and Neuronal Network Encoded by Dorsal Raphe Serotonergic Neurons. Neuropsychopharmacology 41(5) 1404-15. Pubmed ID:26383016
- Ray RS et al (2011) Impaired respiratory and body temperature control upon acute serotonergic neuron inhibition. Science 333(6042) 637-42. Pubmed ID:21798952
- Robinson S et al (2014) Chemogenetic silencing of neurons in retrosplenial cortex disrupts sensory preconditioning. J Neurosci 34(33) 10982-8. Pubmed ID:25122898
- Blum ID et al (2014) A highly tunable dopaminergic oscillator generates ultradian rhythms of behavioral arousal. Elife 3 Pubmed ID:25546305
- Vazey EM et al (2014) Designer receptor manipulations reveal a role of the locus coeruleus noradrenergic system in isoflurane general anesthesia. Proc Natl Acad Sci U S A 111(10) 3859-64. Pubmed ID:24567395
- Akhmedov D et al (2017) Gs-DREADD Knock-In Mice for Tissue-Specific, Temporal Stimulation of Cyclic AMP Signaling. Mol Cell Biol 37(9) Pubmed ID:28167604
- Grace PM et al (2016) Morphine paradoxically prolongs neuropathic pain in rats by amplifying spinal NLRP3 inflammasome activation. Proc Natl Acad Sci U S A 113(24) E3441-50. Pubmed ID:27247388
- DREADD users blog (chemogenetic.blogspot.com/) Accessed July ‘17